Molecular mechanisms of the Bcl-2 proteins
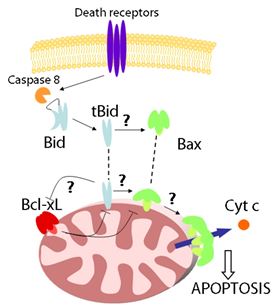 The proteins of the Bcl-2 family are essential regulators of apoptosis.
They control a key event in apoptosis that is the permeabilization
of the mitochondrial outer membrane (MOM). This leads to the release
of the so-called apoptotic factors (like cytochrome c (cyt c), AIF
or Smac/DIABLO) into the cytosol, which induces the activation of caspases
and cell death. Because of the essential role of apoptosis during development,
tissue homeostasis or functioning of the immune system, the regulation
of apoptosis is a fundamental question in biology. In addition, since
the Bcl-2 proteins have an important role in tumorigenesis and in the
cellular responses to anti-cancer therapies, understanding their functioning
is of great therapeutic interest.
The proteins of the Bcl-2 family are essential regulators of apoptosis.
They control a key event in apoptosis that is the permeabilization
of the mitochondrial outer membrane (MOM). This leads to the release
of the so-called apoptotic factors (like cytochrome c (cyt c), AIF
or Smac/DIABLO) into the cytosol, which induces the activation of caspases
and cell death. Because of the essential role of apoptosis during development,
tissue homeostasis or functioning of the immune system, the regulation
of apoptosis is a fundamental question in biology. In addition, since
the Bcl-2 proteins have an important role in tumorigenesis and in the
cellular responses to anti-cancer therapies, understanding their functioning
is of great therapeutic interest.
The Bcl-2 proteins are classified into three subgroups: i) antiapoptotic
proteins, like Bcl-2 or Bcl-xL; ii) proapoptotic proteins like Bax
or Bak that are likely to participate directly in MOM permeabilization;
iii) and the BH3-only proteins, including Bid or Bim, which act like
sensors for the different apoptotic stimuli and initiate apoptosis.
Under normal conditions, many of the proteins of the Bcl-2 family remain
in an apoptosis-inactive form in the cytosol or associated to the outer
mitochondrial membrane. In the presence of apoptotic stimuli, the BH3-only
proteins are activated first. They are considered the initial sensors
that recognize the diverse apoptotic signals in the cell. For example,
Bid is activated by proteolytic cleavage by caspase-8. Once induced
or activated, the Bcl-2 proteins insert into the MOM, where they engage
with other members of the family to regulate cytochrome c release and
cell death.
Despite their high degree of homology, the reasons why the members
of the Bcl-2 family have opposite functions are still obscure. Also,
how the different members of the family engage with each other to decide
whether or not apoptosis is induced needs better understanding. Another
intriguing step in the pathway is their translocation to the mitochondrial
membrane, since the role of the membrane environment on their function
is unknown. Because of these and other questions, the mechanism of
action of the Bcl-2 proteins remains unclear. Our research focuses
on the role of the membrane and of specific lipids, and on the interaction
network between the Bcl-2 members that decides whether or not apoptosis
is induced.
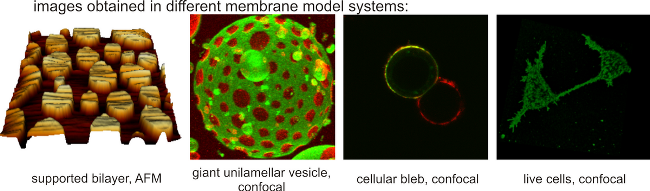
Model membranes and optical microscopy
To investigate dynamic processes in biological membranes we use model
systems of different complexity, ranging from pure lipid bilayers to
cultured cells. A common feature of these membrane systems is that they
can be visualized with optical microscopy and, as a consequence, they
can be used for experiments of time-lapse microscopy, FRAP, FRET, FCS
and other advanced microscopy techniques.
Fluorescence correlation spectroscopy (FCS)
FCS is a technique with single molecule sensitivity that analyzes
the fluctuations in fluorescence intensity within a tiny volume (in
the order of fL). It can be used to measure diffusion coefficients,
fluorophore concentrations, particle sizes, chemical reactions, conformational
changes and binding/unbinding processes, among other. All this makes
FCS an excellent technique for the investigation of dynamic processes.
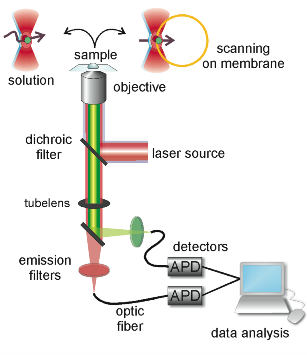
Two-color FCS or fluorescence cross-correlation spectroscopy (FCCS)
is a variant of FCS extremely convenient for the study of molecular
interactions, both in vitro and in vivo. It measures
dynamic co-localization and therefore can be used for binding or dissociation
studies. In this case, two particles of interest are labeled with spectrally
different fluorophores and excited with overlapping laser beams. If
the labeled species interact, they will diffuse together through the
focal volume, inducing simultaneous fluorescence fluctuations and positive
cross-correlation.
In the case of membrane applications of FCS, some issues related to
the sample characteristics delayed the standardization of the technique.
A very interesting approach is scanning FCS (SFCS), which refers to
a group of FCS variants in which the detection volume is scanned through
the sample. They can be classified in strategies where the scan path
is on the membrane or where the beam is scanned across the membrane.
In the first case, the residence time of the fluorophores in the focal
volume is shortened, so that photo-bleaching decreases. It also increases
statistical accuracy due to parallel acquisition. In such an approach,
information about the scanning speed substitutes the calibration of
the detection volume and absolute diffusion coefficients and area concentrations
of the fluorophores are obtained. In the second strategy the laser
beam illuminates the membrane only when it passes through and longer
acquisition times are possible. This is especially relevant in the
case of slow diffusing species typical of lipid membranes and importantly,
it corrects for membrane movements.
Cross-talk between dying and bystander cells in vitro and in vivo: an optogenetics approach
Currently available tools do not allow studying the cross-talk between dying and bystander cells in a spatially and temporally resolved manner. The project aims to develop optogenetically regulated systems allowing the precisely controlled induction of different forms of cell death at a single cell level and use them to study the cross-talk between dying and bystander cells in vitro and in vivo. By employing selective light-controlled activation of different death effectors, this project will study how the different types of cell death shape tissue responses and the underlying mechanisms.
Structural and functional study of mixed lineage kinase domain-like proteins in plants and animals
The discovery of a new protein family in seed plants that is structurally and functionally homologous to animal Mixed lineage kinase domain-like (MLKL) protein, the executioner of necroptosis in animal cells, provided evidence that similar mechanisms may induce plasma membrane permeabilisation in plants and animals. This project aims to understand the molecular principles of MLKL-mediated cell death and immunity by determining and comparing the nanoscale organisation and cellular dynamics of this protein family in plant and animal cells.
Effect of Bcl-2 proteins on mitochondrial structure, dynamics and function
The proteins of the Bcl-2 family are critical regulators of the mitochondrial pathway of apoptosis. Besides this function, evidence suggests that the Bcl-2 proteins have additional functions on several mitochondrial processes which, together with the molecular mechanisms involved, remain poorly understood. The main goal of this proposal is to dissect the differential role of the Bcl-2 proteins in mitochondrial organization and function and to uncover the underlying molecular mechanisms.
Membrane alterations in ferroptosis: from lipid oxidation to pore-formation
Ferroptosis is a caspase-independent form of regulated necrosis characterized by the generation of iron-dependent lipid peroxides in cellular membranes. While it is still unknown how lipid peroxidation leads to ferroptotic cell lysis and death, plasma membrane rupture releases pro-inflammatory damage-associated molecular patterns (DAMPs) leading to necroinflammation and activation of the innate immune system. In this context, ferroptosis has been linked to diseases such as ischemia/reperfusion injury, tissue damage and organ demise, neurodegenerative diseases and cancer. Elucidation of the molecular mechanisms governing membrane rupture during ferroptosis is therefore not only of biological, but also of medical relevance.The overarching goal of this project is to understand how lipid peroxidation triggers plasma membrane permeabilization leading to cell death. We previously found that the final step of ferroptosis execution involves the opening of nanopores at the plasma membrane that cause sustained high cytosolic calcium and cell swelling prior to cell death. Building on these results and our expertise on characterizing membrane permeabilization mechanisms in regulated cell death, in the first aim of this proposal we will determine the alterations in the biophysical properties of cellular membranes in ferroptotic cells. We will use advanced microscopy and biophysical tools to examine the changes in their permeability, fluidity and lateral organization, transmembrane asymmetry and mechanical properties. In the second aim, we will identify by lipidomic analysis which peroxidized lipid species and derivatives are generated in subcellular membranes during ferroptotic progression. Finally, in the third aim, we will validate the functional relevance of previously known and newly identified lipid species for ferroptotic death and relate them to the membrane alterations identified in the first aim. The expected outcome of this research will advance our molecular understanding of ferroptosis by establishing a mechanistic link between lipid peroxidation and execution of cell death.
Mechanisms of MLKL in necroptosis: from intramolecular rearrangements to isoform regulation
Necroptosis is a newly discovered form of regulated necrosis that is inflammatory and plays a role in a number of diseases associated with chronic inflammation and infection. It results in release of the cellular contents after plasma membrane permeabilization dependent on the pseudokinase mixed lineage kinase domain-like (MLKL) protein, which is the most terminal effector of necroptosis known to date. Additionally, non-deadly roles of MLKL in intracellular vesicle trafficking that seem to counterbalance necroptotic cell death have been recently described. However, the molecular mechanism behind these MLKL functions remains obscure and is a matter of debate. In preliminary work, we have discovered that the different isoforms of mouse and human MLKL identified by transcriptome analysis exhibit distinct death-inducing potency. While lack of the inhibitory pseudokinase domain renders MLKL intrinsically active, insertion of just eight amino acids in the C-terminal helix abolishes its necroptotic activity. The main goal of this project is to shed new light on the molecular mechanism how MLKL executes necroptosis. To this aim, we will uncover the structural elements that regulate MLKL activity and determine the contribution of the different MLKL isoforms to MLKL deadly and non-deadly functions. Based on the effect of small differences in the sequence of the C-terminal helix on MLKL activity, we plan to investigate how MLKL structure regulates function. We will then use this information to discover new small molecules that regulate MLKL activity. Finally, we will examine the role of the different MLKL isoforms on the regulation of necroptosis and of additional non-deadly functions. This multidisciplinary project combines cell biology and advanced microscopy with molecular dynamic simulations to disclose new molecular steps involved in the coordination of necroptosis. The identification of new small molecules and proteins implicated in necroptosis regulation will pave the way for the design of new drugs that modulate MLKL activity for human health.
Stoichiometry of homo- and hetero-complexes of Bcl-2 proteins at the single molecule level
The proteins of the Bcl-2 family form a complex interaction network that determines the permeabilization of the mitochondrial outer membrane (MOM) in apoptosis. However, the molecular mechanism how the multiple interactions between Bcl-2 proteins are integrated to control MOM permeabilization remains one of the key questions in the field.The main objective of this project is to uncover the stoichiometry of homo- and hetero-complexes of Bcl-2 proteins in the membrane environment and to examine how protein concentration, phosphorylation, membrane curvature or other family members, regulate these complexes. We aim to achieve unprecedented detail thanks to the use of tailored single molecule techniques, which will provide quantitative information about binding affinity, structural organization, temporal and population heterogeneities, and direct visualization of individual complexes. Extending on our previous single molecule studies of Bax, we will address the following concrete questions:1) Role of protein concentration and membrane curvature in the distribution of Bax oligomeric species. 2) Stoichiometry and lifetime of cBid/Bax and Bcl-xL/Bax complexes in the membrane.3) Relative contribution of Bax auto-activation vs. activation by cBid in the final distribution of Bax oligomeric species. Effect of Bcl-xL on the process.4) Stoichiometry of Bok oligomers in the membrane and regulation by cBid and Bcl-xL, as well as the inhibitory mechanism of Bfl1.5) Affinity between Bcl-2 and PUMA, Bim or tBid, and role of phosphorylation on the interaction between these proteins and in apoptosis regulation. 6)Structural conformation of Bim alone and in complex with DLC1, Bax or Bcl-xL in the membrane environment.The outcome of this research will provide a detailed molecular description of the homo- and heterocomplexes of Bcl-2 proteins (including their regulation) that improves current mechanistic models and brings us closer to a functional understanding of how mitochondrial apoptosis is orchestrated. Given the implication of the Bcl-2 family in several diseases, the outcome of our research will also be important to design new ways of therapeutic intervention targeting the Bcl-2 family and improve the existing ones.
Interplay between the Bcl-2 proteins and Drp1: a biophysical characterization
A key step in the signaling pathway of apoptosis is MOM permeabilization, which is considered the point-of-no-return in the cells commitment to death. Bax is a protein of the Bcl-2 family that directly mediates MOM permeabilization. The machinery for mitochondrial fusion and fission has been shown to also play a role in the regulation of MOM permeabilization, likely by affecting the function of Bax, and vice versa. Drp1 is a large GTPase of the dynamin family that is necessary for mitochondrial division in mammals. During apoptosis, mitochondria undergo massive fragmentation accompanying MOM permeabilization. Interestingly, Bax and Drp1 colocalize at discrete MOM foci that likely correlate with mitochondria/ER contact sites. However, the molecular details and functional consequences of the interplay between Bax and Drp1 remain unresolved.
The main objective of this project is to uncover the molecular mechanism of the interaction between Bax and Drp1 and to correlate it with cellular function. To this aim, we will used functional and single molecule microscopy to understand the structural organization of Bax/Drp1 complexes, including their binding interfaces, and to shed light on the functional consequences of the interaction.
Role of apoptotic foci in mitochondrial outer membrane permeabilisation
Apoptotic cell death regulation is essential for development, immune function and tissue homeostasis. Mitochondrial outer membrane permeabilisation (MOMP) is central for apoptosis execution, and understanding the mechanisms mediating MOMP is crucial for the potential clinical application of apoptosis. Recent studies from the host laboratory found that Bax and Bak protein dimers assemble at specific apoptotic foci to mediate MOMP. The EU-funded APOSITE project will investigate the composition, dynamics and structure of apoptotic foci to elucidate their integral role in apoptosis regulation. Researchers will capitalise on their expertise in cell death and advanced imaging technologies. The multidisciplinary approach promises to solve the question of how Bax and Bak mediate MOMP and to help develop apoptosis modulators for medical applications.

 The proteins of the Bcl-2 family are essential regulators of apoptosis.
They control a key event in apoptosis that is the permeabilization
of the mitochondrial outer membrane (MOM). This leads to the release
of the so-called apoptotic factors (like cytochrome c (cyt c), AIF
or Smac/DIABLO) into the cytosol, which induces the activation of caspases
and cell death. Because of the essential role of apoptosis during development,
tissue homeostasis or functioning of the immune system, the regulation
of apoptosis is a fundamental question in biology. In addition, since
the Bcl-2 proteins have an important role in tumorigenesis and in the
cellular responses to anti-cancer therapies, understanding their functioning
is of great therapeutic interest.
The proteins of the Bcl-2 family are essential regulators of apoptosis.
They control a key event in apoptosis that is the permeabilization
of the mitochondrial outer membrane (MOM). This leads to the release
of the so-called apoptotic factors (like cytochrome c (cyt c), AIF
or Smac/DIABLO) into the cytosol, which induces the activation of caspases
and cell death. Because of the essential role of apoptosis during development,
tissue homeostasis or functioning of the immune system, the regulation
of apoptosis is a fundamental question in biology. In addition, since
the Bcl-2 proteins have an important role in tumorigenesis and in the
cellular responses to anti-cancer therapies, understanding their functioning
is of great therapeutic interest. 